Esters: The Unsung Heroes of The Single Barrel
By Andrew Wiehebrink
“No two barrels are exactly alike.”
This is an often-used phrase that you have likely heard if you have ever had the opportunity to do a private barrel selection. While intended to be a more encompassing statement, this phrase often gets taken quite literal. In other words, most folks would say that no two whiskeys are identical because no two individual barrels are created exactly the same. It seemingly isolates the oak as the one and only culprit for differences in the overall flavor profile.
All other things equal, there are several reasons why whiskey in one barrel might be very different than that of the whiskey in the barrel sitting right next to it. For the purpose of this post, I would like to direct our attention away from the oak itself and concentrate on one such chemical reaction that occurs within the barrel. One that we have very little control over… esterification.
Esterification, or the process of producing esters, occurs when an alcohol runs into an acid while inside the barrel. Esters are fascinating little compounds in the sense that they are responsible for some of the more distinguishable and potent aromas that most of us run into on a frequent basis, not just in our glass of favorite whiskey or wine. The table below outlines some of those esters and their respective aromas you might encounter.
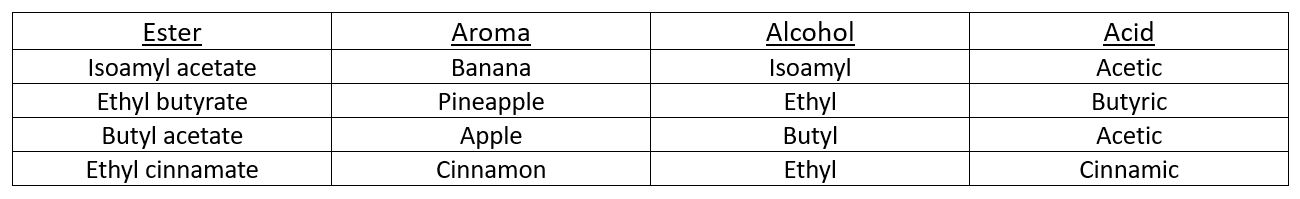
The rate and sequence in which esters are formed is not consistent from barrel to barrel. This is dependent on many factors. Some of the processes are even reversible. No matter how much time has passed, esterification is not static. Meaning this process is happening at varying rates throughout the life cycle of the maturation. Not all esters are created in equal concentrations and sometimes they do not even appear within a given barrel. The formation of esters is going to depend on what type of acids and alcohols are in the distillate itself. This of course is going to vary with different yeast strains, fermentation times/temperatures, and different distillation techniques/equipment.
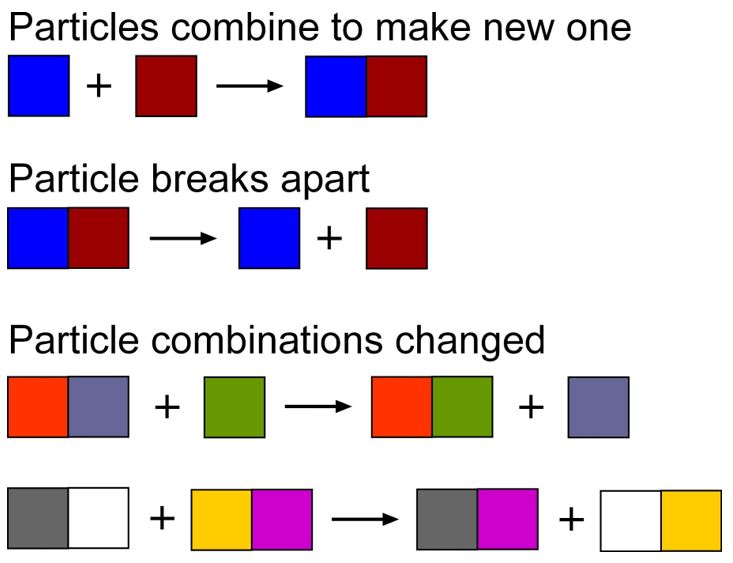
Complicating things further, once an ester is formed, they don’t necessarily have to stay that way for the remainder of the maturation process.

As we see from the graphic above, through a process called transesterification, we can take our existing ester, combine it with an alcohol (usually ethyl) and create a different alcohol and a different ester. To summarize these complications visually, we end up with something like the following:
Previous research1 has shown that esters are near the top of the list in terms of components that have the largest effect on the overall flavor profile of whiskey. As the maturation cycle continues, the concentration of acids and esters will continue to rise2 and become increasingly more influential in the overall flavor profile. We can therefore conclude, that the longer the barrel matures, the greater potential for flavor differentiation between adjacent barrels- but only to a certain extent. Andreasen and Black’s research also showed that throughout the maturation cycle, the concentration of barrel extractives continues to rise. Up to a certain point this isn’t an issue for the esters because, in general, the esters collectively have a lower threshold of perception than do the barrel extractives. However, after an extended period of time in barrel, and depending on the warehouse conditions, the rate at which extractives are pulled could begin to overwhelm the esters and the esters become less impactful, and therefore, flavor profiles differentiate less. (E.g., after 30 years in a barrel, everything just tastes like wood.)
In summary, it is possible that two barrels sitting next to each other in a rickhouse will have flavor differences that are for the most part, undetectable. However, from the explanations and examples shown above, you now know that it is ultimately determined by the creation and manipulations of those esters throughout the majority of the maturation cycle- the unsung heroes of the single barrel.
Cheers!
Andrew Wiehebrink

1 Salo, P: Odor thresholds and relative intensities of volatile aroma components in an artificial beverage imitating whisky
2 Black, R.A. and A.A. Andreasen: Congener Development

